Biogeochemical Cycle/Nutrient Cycle – Carbon, Nitrogen etc.
FUNCTIONS OF ECOSYSTEM
Table of Contents
- The function of an ecosystem is a broad, vast and complete dynamic system. It can be studied under the following three heads.
-
- Energy flow
- Nutrient cycling (biogeochemical cycles)
- Ecological succession or ecosystem development
-
The concept of Energy Flow has been discussed in previous two post of Food Chain and Food Web and Ecological Pyramids. If you have not read it yet, Click Here to read it. Here, we will now discuss about the Various Nutrient Cycles/Biogeochemical Cycles namely – Carbon Cycle, Nitrogen cycle, Water Cycle, Sulphur Cycle and Phosphorus Cycle of the Ecosystem.
BIOGEOCHEMICAL CYCLE
- Energy flow and nutrient circulation are the major functions of the ecosystem.
- The flow of energy in ecosystems is one-way. In contrast, the nutrients which are needed to produce organic material are circulated round the system. They can be recycled again and again indefinitely.
- Carbon, hydrogen, oxygen, nitrogen and phosphorus as elements and compounds makeup 97% of the mass of our bodies and are more than 95% of the mass of all living organisms. These substances are known as macronutrients and form the basis of fats, carbohydrates and proteins.
- In addition to these, about 15 to 25 other elements are needed in some form for the survival and good health of plants and animals. Such as magnesium, Sulphur and phosphorus are needed in minute amounts and are known as micronutrients.
- These elements or mineral nutrients are always in circulation moving from non-living to living and then back to the non-living components of the ecosystem in a more or less circular fashion. Nutrient cycles has a reservoir pool, which is a large, slow-moving non-biological component, and an exchange pool, which is a smaller, more active portion where the nutrient is exchanged between biotic and abiotic parts of the ecosystem.
- This circular fashion is known as Biogeochemical Cycle (bio for living; geo for atmosphere).
- Among the most important nutrient cycles are the carbon nutrient cycle and the nitrogen nutrient cycle.
- There are many other nutrient cycles that are important in ecology, including a large number of trace mineral nutrient cycles.
Types of Nutrient Cycles
- Based on the replacement period, a Nutrient Cycle is referred to as Perfect or Imperfect cycle.
- A perfect Nutrient Cycle/Biogeochemical Cycle is one in which nutrients are replaced as fast as they are utilised. Most gaseous cycles are generally considered as perfect cycles.
- In contrast sedimentary cycles are considered relatively imperfect, as some nutrients are lost from the cycle and get locked into sediments and so become unavailable for immediate cycling.
- Based on the nature of the reservoir, a Nutrient Cycle/Biogeochemical Cycle is referred to as Gaseous or Sedimentary cycle
- Gaseous Cycle: the reservoir is the atmosphere or the hydrosphere— water cycle, carbon cycle, nitrogen cycle, etc. and
- Sedimentary Cycle: the reservoir is the earth’s crust(soluble elements mostly found in earth’s crust) — phosphorous cycle, sulphur cycle, calcium cycle, magnesium cycle
Gaseous Cycles
The most important gaseous Biogeochemical Cycle includes – water, carbon and nitrogen.
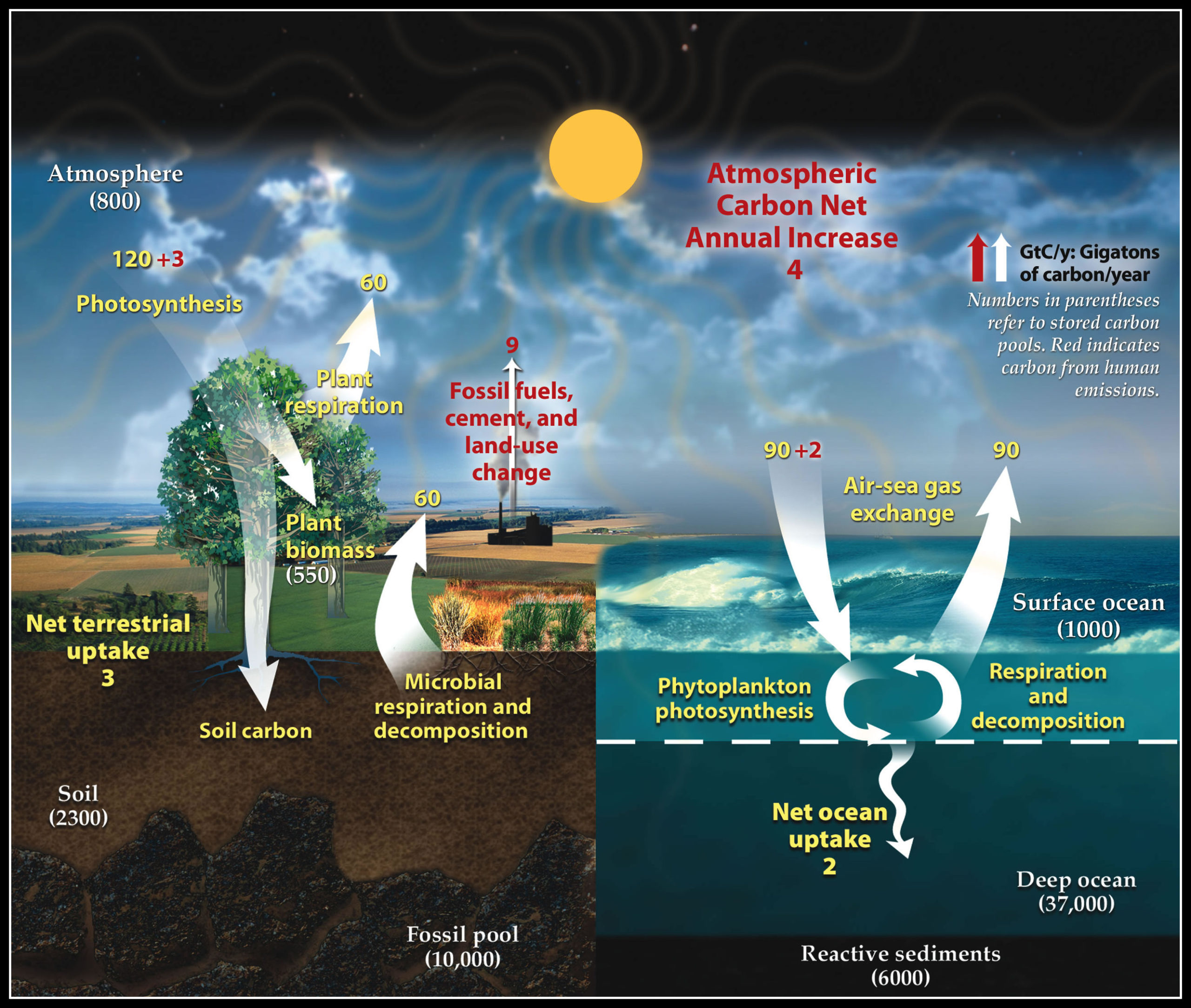
CARBON CYCLE (GASEOUS CYCLE)
-
- Carbon is a minor constituent of the atmosphere as compared to oxygen and nitrogen.
- However, without carbon dioxide life could not exist because it is vital for the production of carbohydrates through photosynthesis by plants.
- It is the element that anchors all organic substances from coal and oil to DNA.
- Carbon is present in the atmosphere, mainly in the form of carbon dioxide (CO2).
-
- Carbon cycle involves a continuous exchange of carbon between the atmosphere and organisms. Carbon cycle is mainly the conversion of carbon dioxide.
- This conversion is initiated by the fixation of carbon dioxide from the atmosphere through photosynthesis. Carbon from the atmosphere moves to green plants by the process of photosynthesis, and then to animals.
- Such conversion results in the production of carbohydrate, glucose that may be converted to other organic compounds. Some of the carbohydrates are utilised directly by the plants itself.
- During the process, more Carbon dioxide is generated and is released through its leaves or roots during the day.The remaining carbohydrates not being utilised by the plant become part of the plant tissue.
- Plant tissues are either being eaten by the herbivorous animals or get decomposed by the microorganisms.
- The herbivores convert some of the consumed carbohydrates into carbon dioxide for release into the air through respiration.
- The micro-organisms decompose the remaining carbohydrates after the animal dies.
- By process of respiration and decomposition of dead organic matter, it returns to the atmosphere. It is usually a short term cycle.
- Some carbon also enters a long term cycle. It accumulates as un-decomposed organic matter in the peaty layers of marshy soil or as insoluble carbonates in bottom sediments of aquatic systems which take a long time to be released.
- In deep oceans, such carbon can remain buried for millions of years till geological movement may lift these rocks above sea level.
- These rocks may be exposed to erosion, releasing their carbon dioxide, carbonates and bicarbonates into streams and rivers.
- Fossil fuels such as coals, oil and natural gas etc. are organic compounds that were buried before they could be decomposed and were subsequently transformed by time and geological processes into fossil fuels. When they are burned the carbon stored in them is released back into the atmosphere as carbon dioxide.
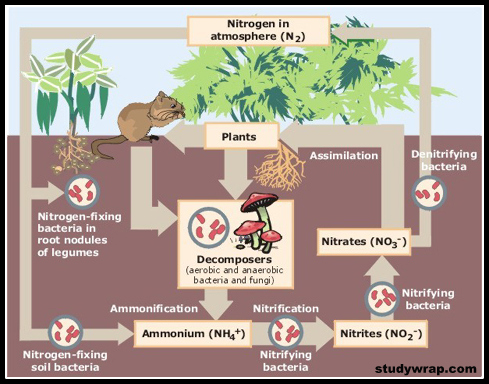
NITROGEN CYCLE (GASEOUS CYCLE)
-
- Apart from carbon, hydrogen and oxygen, nitrogen is the most dominant element in living organisms as it is constituent of amino acids, proteins, hormones, chlorophylls and many of the vitamins.
- Plants compete with microbes for the limited nitrogen that is available in the soil. Thus, nitrogen is a limiting nutrient for both natural and agricultural ecosystems.
- Nitrogen exists as two nitrogen atoms (N2) in the atmosphere.
- In nature, lightning and ultraviolet radiation provide enough energy to convert nitrogen to nitrogen oxides (NO, NO2, N2O).
- Industrial combustions, forest fires, automobile exhausts and power-generating stations are also sources of atmospheric nitrogen oxides.
Nitrogen Fixing – Nitrogen to Ammonia (N2 to NH3)
-
-
- There is an inexhaustible supply of nitrogen in the atmosphere, but the elemental form cannot be used directly by most of the living organisms.
- Nitrogen needs to be ‘fixed’, that is, converted to ammonia, nitrites or nitrates, before it can be taken up by plants.
- Nitrogen fixation on earth is accomplished in three different ways
- By microorganisms (bacteria and blue-green algae),
- By man using industrial processes (fertiliser factories) and
- To a limited extent by atmospheric phenomenon such as thunder and lighting.
- Certain microorganisms are capable of fixing atmospheric nitrogen into ammonia and ammonium ions.
- The enzyme, nitrogenase which is capable of nitrogen reduction is present exclusively in prokaryotes. Such microbes are called N2-fixers. These include:
- free-living nitrogen fixing bacteria (non-symbiotic nitrogen-fixing bacteria or nitrogen-fixing soil bacteria) (e.g. aerobic Azotobacter and anaerobic Clostridium),
- symbiotic nitrogen-fixing bacteria (e.g. Rhizobium) living in association with leguminous plants and non-leguminous root nodule plants and
- Some cyanobacteria (a major source of nitrogen fixation in oceans) (blue-green algae. E.g. Nostoc, Anabaena, Spirulina).
-
Nitrification – Ammonia to Nitrates
-
-
- Ammonium ions can be directly taken up as a source of nitrogen by some plants.
- Others absorb nitrates which are obtained by oxidising ammonia and ammonium ions.
- Ammonia and ammonium ions are oxidised to nitrites or nitrates by two groups of specialized bacteria.
- Ammonium ions are first oxidised to nitrite by the bacteria Nitrosomonas and/or Nitrococcus.
- The nitrite is further oxidized to nitrate with the help of the bacterium Nitrobacter.
- These steps are called These nitrifying bacteria are chemoautotrophs (they use inorganic chemical energy sources to synthesise organic compounds from carbon dioxide).
- The nitrate thus formed is absorbed by plants and is transported to the leaves.
- In leaves, it is reduced to form ammonia that finally forms the amine group of amino acids, which are the building blocks of proteins. These then go through higher trophic levels of the ecosystem.
- Nitrification is important in agricultural systems, where fertiliser is often applied as ammonia.
- Conversion of this ammonia to nitrate increases nitrogen leaching because nitrate is more water-soluble than ammonia.
-
Ammonification – Urea, Uric Acid to Ammonia
-
-
- Living organisms produce nitrogenous waste products such as urea and uric acid (organic nitrogen).
- These waste products, as well as dead remains of organisms, are converted back into inorganic ammonia and ammonium ions by the bacteria. This process is called ammonification.
- Some of this ammonia volatilises and re-enters the atmosphere, but most of it is converted into nitrate by soil bacteria.
-
Denitrification – Nitrate to Nitrogen
-
-
- Nitrate present in the soil is reduced to nitrogen by the process of denitrification.
- In the soil as well as oceans there are special denitrifying bacteria (Pseudomonas and Thiobacillus), which convert the nitrates/nitrites to elemental nitrogen.
- This nitrogen escapes into the atmosphere, thus completing the cycle.
-
| Step 1: N2 Fixing Nitrogen → Ammonia or Ammonium Ions
Step 2: Nitrification Ammonia or Ammonium Ions → Nitrite → Nitrate Step 3: Ammonification Dead Matter + Animal Waste (Urea, Uric Acid) → Ammonia or Ammonium Ions Most of the ammonia escapes into the atmosphere. Rest is Nitrified (Step 2) to nitrates. Some of the nitrates is available for plants. Rest is Denitrified (Step 4). Step 4: Denitrification Nitrate → Nitrogen |
-
- The amount of nitrogen fixed by man through the industrial process has far exceeded the amount fixed by the Natural Cycle.
- As a result, nitrogen fixed by man has become a pollutant which can disrupt the balance of nitrogen. It may lead to Acid rain, Eutrophication and Harmful Algal Blooms.
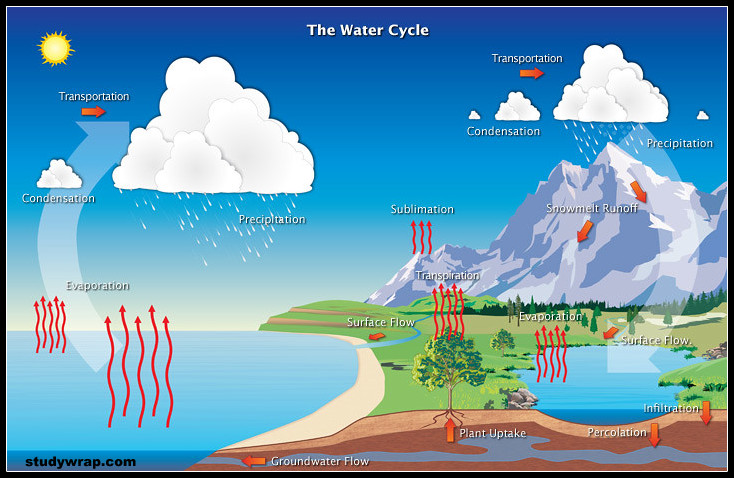
WATER CYCLE
-
- Water undergoes a cycle from the ocean to land and land to ocean.
- The hydrological cycle describes the movement of water on, in, and above the earth which is driven by solar energy.
- The distribution of water on earth is quite uneven. Many locations have plenty of water while others have very limited quantity.
- The hydrological cycle is the circulation of water within the earth’s hydrosphere in different forms i.e. the liquid, solid and the gaseous phases.
-
- Water moves from one reservoir to another by the processes of evaporation, transpiration, condensation, precipitation, deposition, runoff, infiltration and groundwater flow.
- About 71 % of the planetary water is found in the oceans. The remaining is held as freshwater in glaciers and icecaps, groundwater sources, lakes, soil moisture, atmosphere, streams and within life.
- Nearly 59 % of the water that falls on land returns to the atmosphere through evaporation from over the oceans as well as from other places. The remainder runs-off on the surface, infiltrates into the ground or a part of it becomes glacier.
- Cycling of all other nutrients is also dependent upon water as it provides their transportation during the various steps. It acts as a solvent medium for their uptake of nutrients by organisms.
Sedimentary Cycle
Phosphorus, calcium and magnesium circulate by means of the Sedimentary Biogeochemical Cycle.
The element involved in the Sedimentary Biogeochemical Cycle follows a basic pattern of flow through erosion, sedimentation, mountain building, volcanic activity and biological transport.
PHOSPHORUS CYCLE (SEDIMENTARY CYCLE)
-
- Phosphorus plays a central role in aquatic ecosystems and water quality.
- Unlike carbon and nitrogen, which come primarily from the atmosphere, phosphorus occurs in large amounts as a mineral in phosphate rocks and enters the cycle from erosion and mining activities.
- This is the nutrient considered to be the main cause of excessive growth of rooted and free-floating microscopic plants in lakes which leads to eutrophication. Phosphates in the soil are taken into plants for protein synthesis and are passed through the food chains of ecosystems.
-
- The main storage for phosphorus is in the earth’s crust. On land, phosphorus is usually found in the form of phosphates.
- By the process of weathering and erosion, phosphates enter rivers, streams and finally oceans.
- In the ocean, phosphorus accumulates on continental shelves in the form of insoluble deposits.
- After millions of years, the crustal plates rise from the seafloor and expose the phosphates on land.
- After more time, weathering will release them from rock, and the cycle’s geochemical phase begins again.
- The phosphorus cycle can be easily disrupted by the use of phosphate fertilisers in modern agriculture.
- Most manufactured phosphate fertilisers are produced from phosphate rocks but are rapidly lost from the exchange pool to marine deposits as they are easily leached from the soil.
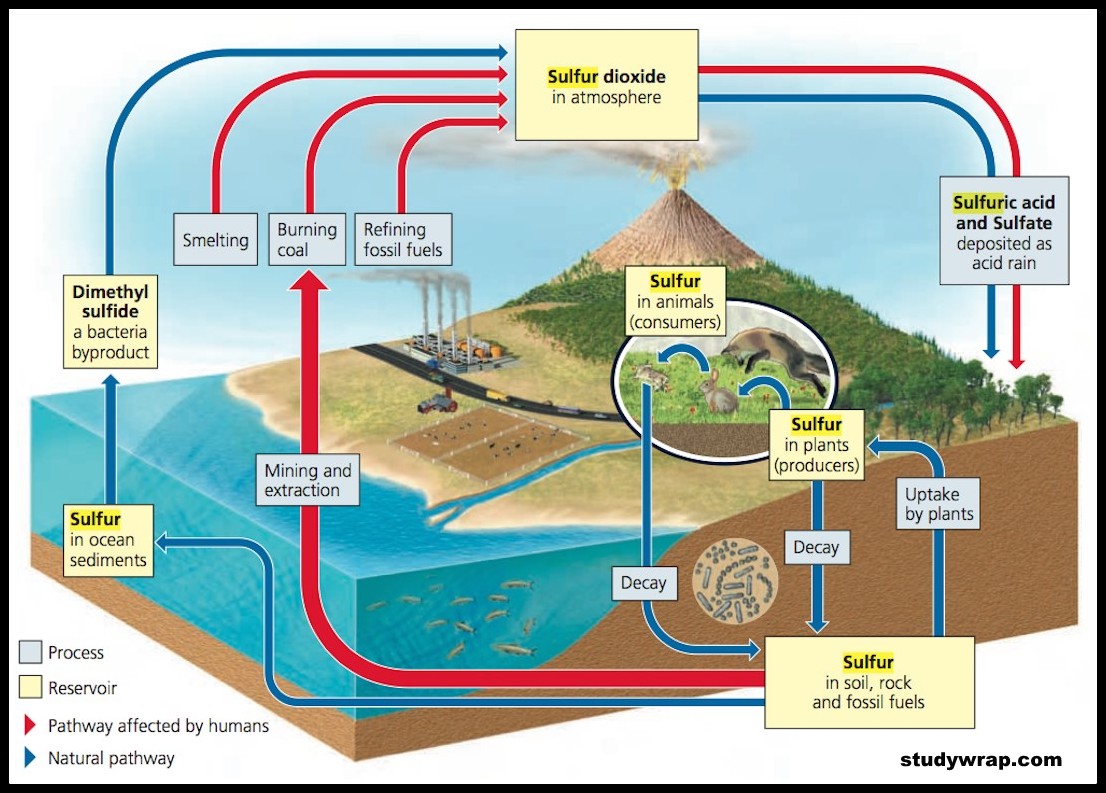
SULPHUR CYCLE (SEDIMENTARY CYCLE)
-
- The sulphur reservoir is in the soil and sediments where it is locked in organic (coal, oil and peat) and inorganic deposits (pyrite rock and sulphur rock) in the form of sulphates, sulphides and organic sulphur.
- It is released by weathering of rocks, erosional runoff and decomposition of organic matter and is carried to terrestrial and aquatic ecosystems in salt solution.
-
- The sulphur cycle is mostly sedimentary except two of its compounds, hydrogen sulphide (H2S) and sulphur dioxide (SO2), which add a gaseous component.
- Sulphur enters the atmosphere from several sources like volcanic eruptions, combustion of fossil fuels (coal, diesel etc.), from the surface of the ocean and gases released by decomposition.
- Atmospheric hydrogen sulphide also gets oxidised into sulphur dioxide.
- Atmospheric sulphur dioxide is carried back to the earth after being dissolved in rainwater as weak sulphuric acid (acid rain).
- Whatever the source, sulphur in the form of sulphates is taken up by plants and incorporated through a series of metabolic processes into sulphur bearing amino acid which is incorporated in the proteins of autotroph tissues. It then passes through the grazing food chain.
- Sulphur bound in a living organism is carried back to the soil, to the bottom of ponds and lakes and seas through excretion and decomposition of dead organic material.
Other Mineral Cycles
-
- Other than carbon, oxygen, nitrogen etc. being the principal geochemical components of the biosphere, many other minerals also occur as critical nutrients for plant and animal life.
- They usually occur as salts dissolved in soil water or lakes, streams and seas.
- Mineral salts come directly from the earth’s crust by weathering where the soluble salts enter the water cycle, eventually reaching the sea.
- Other salts are returned to the earth’s surface through sedimentation, and after weathering, they again enter the cycle.
- All living organisms fulfil their mineral requirements from mineral solutions in their environments. Other animals receive their mineral needs from the plants and animals they consume.
- After the death of living organisms, the minerals are returned to the soil and water through decomposition and flow.
So, this was all about the Biogeochemical Cycles/ Nutrient Cycles which includes – Carbon Cycle, Nitrogen cycle, Water Cycle, Sulphur Cycle and Phosphorus Cycle.
In the Next Post (Click Here), we will learn about the Ecological Succession, another Function of Ecosystem. Hope you like the post, Do comment your opinion.



Greatly appreciated!